Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
This is it Mohsena weekly newsletter sent every Friday from one of the Verge’s top reviewers Victoria song Which dissects and discusses the latest tools and potions that you swear will change your life. Mohsen It arrives in our subscribers’ inboxes at 10 a.m. ET. Subscribe to Mohsen here.
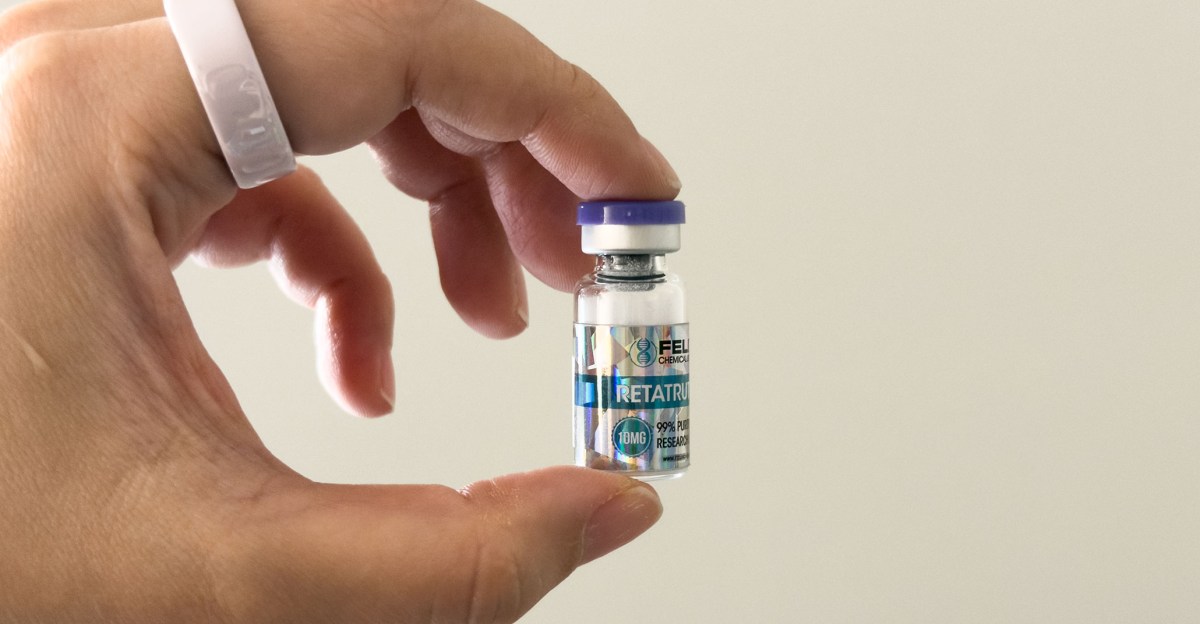
On TikTok, it’s very easy to find videos of influencers reformatting vials of crushed peptides. Most often, influencers carry vials of gray market retatrotide, an unapproved weight-loss drug known colloquially variously as GLP-3 (because it adds two additional stimulants, glucagon and GIP), Reta, and Ratatouille. Other supplies available are alcohol swabs, syringes, and germicidal water. Kitchen chemists say how easy it is to turn a powdery substance in a bottle into a peptide that can be injected into the body. To find out dosages, simply use our online peptide calculator. Storage instructions are everywhere, but generally they boil down to “keep in the refrigerator for 30 to 90 days.”
Most educational programs do not include simple, conclusive instructions such as “wash your hands” or “disinfect all surfaces.” The vast majority of influencers don’t wear rubber gloves. The most anyone would do is run an alcohol swab over the caps of Retrotide water bottles and the germs will germinate.
When I show these clips to a pharmacy that specializes in formulations, her face falls.
For the past month, I’ve been chasing a vial of Retatrotide in the refrigerator.
After searching So-called GLP-3s Last month, I got a bottle through a TikTok influencer’s link tree. The problem with gray market peptides is that you don’t really know what you’re buying. Sites that sell these peptides—whether GLP-1 or others—sometimes say their product is 99 percent pure and post an official-looking certificate of authenticity.
You want to make sure that your seller is the real dealInfluencers say. Look for third-party laboratory certification. This is how you know it’s legit. For the suppliers I use, the link is in my bio and use my code to get 10 percent off.
My hypothesis is that my vial of questionably sourced retatrotide—an unapproved drug still undergoing FDA phase III clinical trials—is not actually retatrotide. At least, not the same one Eli Lilly is using in its clinical trials. I figured I’d consult a pharmacist, find a reputable third-party lab, send over my vial, and bada bing, bada boom, I’d have an answer.
As it turns out, it’s not that easy.
To understand why, we have to step back a little. Typically, if you need medication, you go through your doctor to get a prescription, and the pharmacy dispenses it. In 2022, there was a shortage of GLP-1 drugs, prompting the FDA to allow compounding pharmacies to sell the drugs.
Compounding pharmacies create customized versions of medications tailored to a specific patient when a commercial option is not viable. Suppose you are allergic to an ingredient in the commercial version of the medication you need. Your doctor may direct you to a compounding pharmacy, which will reconstitute the medication and mix it for you (generally). The market has boomed in recent years with GLP-1 drugs such as Ozempic, Wegovy, Mounjaro, and Zepbound. For many people, compounding pharmacies have provided an affordable alternative to obtaining these medications when they are helpful but not available through the traditional route. If you’re not diabetic, for example, but have been struggling to lose weight due to hormonal conditions, you may not qualify under the terms set forth in your health insurance plan. In a Money planet a reportone user found his access denied after a “significant improvement” in using the drug.
For these people, compound drugs were a godsend. But compounding pharmacies it’s not Same thing as the gray market. The gray market consists of any wholesaler, distributor, or manufacturer who sells outside approved distribution networks. Purchasing on the gray market often means reconstructing medications yourself, as TikTokers do. You can “do your homework,” as influencers urge, and still be easily confused between a legitimate compounding pharmacy supplying the original active pharmaceutical ingredients (APIs) with approved testing and licensure, and an unknown source claiming to be of the same composition, quality, and potency.
“You can get the API from other places. It’s not like it’s just being delivered to the Eli Lilly factory or wherever the drug is made. But for something that’s not even approved by the FDA yet, like retatrotide, it’s He is This is stated only for research purposes Annie Lambertpharmacist and formulation specialist at Wolters Kluwer. “In the gray market, it should raise more questions, like ‘What is quality? What is safety? What is purity? How can I know these things? How can I know these things?” Validation These things?”
There’s always a risk of resorting to a combination drug, Lambert says. However, there is far less The risk of going to a compounding pharmacy instead of going to the gray market.
In our call, I showed Lambert the questionable vial of retatrotide. The brand claims 99% purity, but what does that actually mean in this context?
There’s a clear difference between purity and potency, Lambert says. Potency refers to the active ingredient in the correct dosage. A Tylenol tablet is expected to contain about 325 mg of the active ingredient, plus some other inactive ingredients to help the medication bind together. There too University of the South Pacific study – A written document that explains and defines quality standards. There is no study on retatrotide, and the FDA explicitly states that retaret is not an ingredient. any A drug approved by the Food and Drug Administration (FDA) and not on the 503A Bulk Drug Substance List, which details all the bulk ingredients that legitimate compounding pharmacies can use to manufacture drugs.
But purity is a different story. Lambert says purity indicates there are endogenous toxins or exogenous substances in the drug that could cause harm.
“When you’re talking about injecting something into yourself, the level is high,” Lambert says. “There are some acceptable levels of endotoxin. The tolerable level is usually set in the USP study. If it’s not an approved product, that standard is questionable as well.”
So, even if you send the vial to a “reputable third-party lab,” when there is no official study available to refer to, it becomes a matter of how and What Laboratory being tested for. As for gray market peptides, it’s impossible to determine if you’re getting the real deal because a safe, tested, “official” version of retatrotide doesn’t exist yet. Of the listings I’ve seen that claim third-party testing, most only address purity. But as Lambert said, with uncertified products, there is no official agreed-upon standard for “purity.”
I still plan to send this bottle out for testing after more research and thought. But the big question is, what can any result tell me?
While I like to say “Just don’t do it!” Regarding unapproved medications, I am well aware of the feeling of desperation when a medication that could help you is arbitrarily withheld due to red tape and cost. Just this week, my pharmacy called to tell me that my insurance company had rejected the medication I needed, and I now had to jump through several bureaucratic hurdles to get it. A broken healthcare system, insensitive doctors, and the stigma associated with large bodies will make alternative sources of GLP-1 impossible for some people to resist.
So I asked Lambert about the differences between how legitimate compounding pharmacies versus gray market work, and how to spot the difference.
According to Lambert, legitimate compounding pharmacies will conduct testing and verification of raw ingredients or APIs. Once the medicine is formulated, sterility and endotoxin testing is also performed, among other things, to ensure that the medicine lasts for the duration stated in the instructions and packaging. (Most gray market certificates of authenticity I’ve reviewed only list purity—not these other factors.) Additionally, these standards and processes are imposed by state boards of pharmacy, accrediting bodies, and the FDA itself.
“As a patient, you should always be able to ask, ‘Where does this drug come from?’” Lambert says. For example, if you use a service like Ro for GLP-1s or other medications, these companies source theirs from a smaller compounding pharmacy — they don’t make them themselves. It is the responsibility of consumers to ensure that the pharmacy sending the medication is licensed in the state in which they live. This pharmacy must also be able to provide sterile compound labels or approvals. You should also ask pharmacies to source the APIs and obtain a third-party certificate of analysis for each batch number. Although third-party testing is not required for compounding pharmacies, it is a good idea to ask a question if you feel like something is not quite right.
Other smart questions include:
If you are looking to obtain GLP-1s or retatrutide from a spa, nurse practitioner, or other health facility, it is doubly important to ask where they Sources are obtained from. If they don’t name a valid pharmacy, you may inadvertently get gray market peptides.
My vial of Retatrotide only came with a vial of germicidal water. There were no instructions for reconstitution, no additional materials such as injections, and no storage instructions. When I bought it, I swore it was sold with a “Lab Certification” link and a batch number to verify purity. As of this writing, the seller’s product listing has no such certifications available. However, there is a note that it will last for 56 days in the refrigerator once reconstituted, and up to a year in powder form. (A year ago when not clear.)
I have no plans to remake this bottle. But if I did, I’d love to find better tutorials than the ones I found on TikTok. As Lambert explains, compounding pharmacists do a lot of work in laboratories when compounding drugs.
“At a minimum, according to USP standards, when we gather in direct pharmacy or healthcare settings, I will at least wash my hands, disinfect the counter, disinfect the top of the lid, and you want everything to be as clean as possible,” she says.
“But perhaps the first question to ask is: How much risk am I willing to take?”
Photography by Victoria Song/The Verge